
The initial versions of Computer Assisted Surgery (CAS) and Robotics Assisted Surgery (RAS) were introduced into the operating room (OR) in the late 1980s. Robotic surgery was first performed by Kwoh in 1985 when a standard industrial (Puma 560) robotic arm was used to place a needle for a brain biopsy while being guided by a computed tomography (CT). The robot was relegated to the role of a traditional stereotactic frame in neurosurgery. However, the company that marketed Puma 560 (Westinghouse Limited), refused to allow the robot to be used for surgery purposes on the basis that it was unsafe, since the industrial robot was designed to be used inside a barrier away from all contact with people. Thus, in spite of the encouraging preliminary results which indicated that, compared with a conventional stereotactic frame, the robot could position itself automatically and very accurately, Kwohĺs work was ceased. Later, prostate (soft-tissue surgery in the transurethral resection of the prostrate (TURP) for benign prostatic hyperplasia) surgery was first performed by the Puma 560 industrial robot in 1988.
Also in the mid-to-late 1980ĺs a group of researchers at the National Air and Space Administration (NASA) Ames Research Center working on virtual reality became interested in using this information to develop telepresence surgery. This concept of telesurgery became one of the main driving forces behind the development of surgical robots. In the early 1990ĺs several of the scientists from the NASA-Ames team joined the Stanford Research Institute (SRI). Working with SRIĺs other robotocists and virtual reality experts, these scientists developed a dexterous telemanipulator for hand surgery. One of their main design goals was to give the surgeon the sense of operating directly on the patient rather than from across the room. While these robots were being developed, general surgeons and endoscopists joined the development team and realized the potential these systems had in ameliorating the limitations of conventional laparoscopic surgery.
Several of the surgeons and engineers working on surgical robotic systems for the Army eventually formed commercial ventures that lead to the introduction of robotics to the civilian surgical community. Notably, Computer Motion, Inc. of Santa Barbara, CA used seed money provided by the Army to develop the Automated Endoscopic System for Optimal Positioning (AESOP), a robotic arm controlled by the surgeon voice commands to manipulate an endoscopic camera. Shortly after AESOP was marketed, Integrated Surgical Systems (now Intuitive Surgical) of Mtn. View, CA licensed the SRI Green Telepresence Surgery system. This system underwent extensive redesign and was re-introduced as the Da Vinci surgical system. Within a year, Computer Motion put the Zeus system into production.
Today, many robots and robot enhancements are being researched and developed. Schurr et al., at Eberhard Karls Universityĺs section for minimally invasive surgery have developed a master-slave manipulator system that they call ARTEMIS. This system consists of two robotic arms that are controlled by a surgeon at a control console. Several labs, are designing and developing systems and models for reality-based haptic feedback in minimally invasive surgery and also combining visual servoing with haptic feedback for robot-assisted surgery. In addition to Prodoc, ROBODOC and the systems mentioned above several other robotic systems have been commercially developed and approved by the FDA for general surgical use.
The da Vinci and Zeus systems (see figure below) are similar in their capabilities but different in their approaches to robotic surgery. Both systems are comprehensive master-slave surgical robots with multiple arms operated remotely from a console with video assisted visualization and computer enhancement. In the da Vinci system, which evolved from the telepresence machines developed for NASA and the U.S. Army, there are essentially three components: a vision cart that holds a dual light source and dual three-chip cameras, a master console where the operating surgeon sits, and a moveable cart where two instrument arms and the camera arm are mounted. The camera arm contains dual cameras and the image generated is three-dimensional. The master console consists of an image processing computer that generates a true three dimensional image with depth of field; the view port where the surgeon views the image; foot pedals to control electrocautery, camera focus, instrument/camera arm clutches, and master control grips that drive the servant robotic arms at the patientĺs side. The instruments are cable driven and provide seven degrees of freedom. This system displays its three-dimensional image above the hands of the surgeon so that it gives the surgeon the illusion that the tips of the instruments are an extension of the control grips thus giving the impression of being at the surgical site.
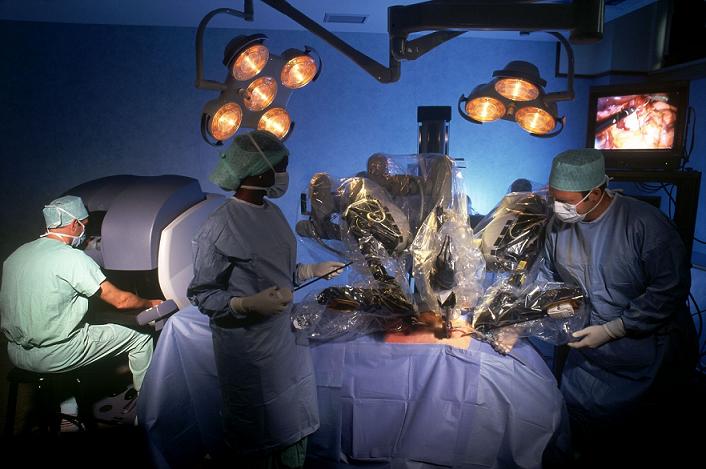
The Zeus system is composed of a surgeon control console and three table-mounted robotic arms. The right and left robotic arms replicate the arms of the surgeon and the third arm is an AESOP voice controlled robotic endoscope for visualization. In the Zeus system, the surgeon is seated comfortably upright with the video monitor and instrument handles positioned ergonomically to maximize dexterity and allow complete visualization of the OR environment. The system uses both straight shafted endoscopic instruments similar to conventional endoscopic instruments and jointed instruments with articulating end-effectors and seven degrees of freedom.
The Different Approaches to Robotic Surgery
1) Supervisor-Controlled Surgical Systems
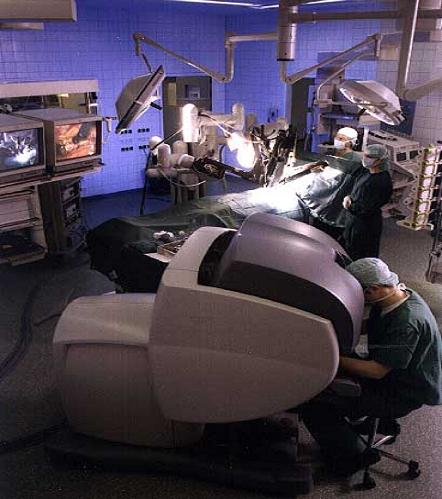
A supervisor-controlled robotic surgical system offers the highest level of automation. It does require a significant amount of preparation to set up to perform each surgery. A specific set of commands, unique to both the patient and the procedure being performed, is first entered into the system by the surgeon and support team. This is accomplished with extensive mapping of the body using three-dimensional medical imaging. Just prior to the surgery, the system is then registered to match the patient's body to the mapping in the surgical system. The surgeon tests the robotĺs motions and places the robot in the appropriate start position. Once the surgery is under way, the robotic system will automatically execute the procedure. The surgeon will observe the procedure intently, and intervene only if necessary (see figure below). The most famous prototype is the ROBODOC system developed by Integrated Surgical Systems, which is commonly used in orthopedic surgeries.

2) Shared Control Robotic Surgical Systems
In this system, the human surgeon does the bulk of the work, actually operating the surgical tools by hand, while the robot assists in performing surgical procedures when needed. The system monitors the activities of the surgeon, providing stability and support to the surgeon's movements using a technique called active constraint. In this technique, the surgeons program the robot to recognize the areas of the surgical field as forbidden, boundary, close and safe. Safe regions are the main focus of the surgery. Close regions border easily damaged soft tissue and the boundary is where soft tissue begins. As the surgeon nears these dangerous areas, the robot pushes back against the surgeon, or in some cases, when the forbidden zone is reached, the robotic system actually locks up to prevent any further injury. Therefore, through forced feedback in the surgical tools, the system limits their use within the appropriate area. As with supervisor-controlled systems, there is some setup required by the surgeon prior to the procedure to define the regions of the surgical field.
3) Telesurgery Systems
With this system, a surgeon actually directs the actions of robotic arms. In effect, the robot becomes an extension of the surgeon. The system consists of surgical arms within a surgical suite and a separate viewing and control console (see figure below). The surgeon views the surgical field in a three-dimensional viewing screen and manipulates the robotic arms from hand-held controls within the console. Another surgeon in the surgical suite changes out tools on the robotic arms as needed during the procedure. Small incisions are made in the body and the tools are then inserted. Once this step is complete, the surgeon can then operate. This technology allows surgeons to make quicker, more controlled and more accurate movements by using the robot arm with its wider range of motions and hence provides a means to perform minimally invasive surgical procedures. The most common variety, the da Vinci Robotic Surgical System, enhances the surgery by providing 3-D visualization deep within hard-to-reach places like the heart, as well as enhancing wrist dexterity and control of tiny instruments. These systems also allow more surgeons to perform these procedures, since many of the techniques performed by robot assistants are highly skilled and extremely difficult for humans to master. Now more procedures (like artery repair and valve repair) can be done without long recovery times or bodily injury.
Telesurgical systems include:
(i) The Da Vinci Surgical System (Approved by FDA in July 2000)
(ii) Zeus Robotic Surgical System
(iii) Aesop Robotic Surgical System (the first robot to be cleared by FDA for assisting surgery in the OR)
4) Robotic Radiosurgery Systems
Robots are also used in delivering radiation for the treatment of tumors. These systems use robotics to control highly focused beams of ionizing radiation to precise locations within the body. Medical imaging first locates the tumor and a map of the area to be treated is created. A series of commands are then entered by the physician into the system to instruct it how to deliver the treatment. The patient is then registered with the system for proper positioning of the body and the treatment is begun. The robot then follows the commands to precisely deliver a series of doses to the tumor. This reduces the risk of damage to surrounding tissues.