Home General Diagnostics Information
Clinical Laboratory Diagnostics
Current Point-of-Care
Diagnostics Current Diagnostics
Companies Future Point-of-Care Diagnostics
References
Current Point-of-Care Diagnostics

Source: www.washington.edu/.../article_gadget.html
Due to the fact that most of the clinical laboratory
errors occur during the preanalytical phase, meaning wrong labels on samples
taken or loss of sample altogether, performing diagnostic analysis on site
would prove a valuable method of choice for error reduction. Using Point-of-Care
technology at one location will virtually eliminate all transportation problems
and reduce the amount of people involved between blood sampling and data
interpretation. Products such as glucose monitoring devices have already
resulted in a dramatic improvement in diabetic patient care and quality of
life, while reducing the taxing demands such frequent tests would have on
clinical laboratories. The primary limitation with POCT in its current state is
the lack of performance analysis to compare its accuracy with that of the
clinical setting. In addition, the current state of POCT technology cannot
analyze as wide of a variety of parameters as can be performed in the
laboratory. In general, one device can test anywhere from 1 to 20 parameters
but to do so may require repeat tests with five or more disposable lab on a
chip cartridges. Currently, it is difficult to compare between POCT and the
central laboratory making it difficult to determine if any technology or
approach is a significant improvement over the other.
Current Limitations
1.
Requires
frequent calibration
2.
Unproven track
record
3.
Long term
storage problematic
4.
Surviving harsh
conditions without failure
5.
Contamination
6.
Limited
diagnostic ability for each device
7.
Lack of technology
8.
Not yet a
complete walk away technology (no personnel supervision)
9.
Currently a
higher cost than most clinical laboratory tests
Possible Advantages
1.
Direct point of
care technology avoids long sample transport
2.
Faster medical
decisions
3.
Portable
4.
Disposable
technology
5.
Potential for
reduced cost
6.
Improved fluid
handling
7.
Sample size
reduction
8.
Hands free
9.
Third world
10.
Biodefense
applications
11.
Personal care
12.
Emergency
response equipment
Analysis techniques
Depending
on the sample to be analyzed and the target analytes or biomarkers, there may
exist a variety of analysis and measurement techniques as shown in table 3
below.
Table 3 Analytical principles used for the determination of analytes
|
Principle of analysis |
Principles of measurement |
Examples |
|
1) Light scatter |
Laser-induced forward scatter |
Blood cell count |
|
2) Electrodes |
Voltage changes |
Electrolytes |
|
3) Chemical |
Optical/transmission |
Creatinine |
|
4) Biochemical assay |
Optical/transmission |
GOT,LDH, CK etc. |
|
5) Antibody-based assays |
Optical/light scattering etc. |
Hormones, drugs |
|
6) Electrochemical based assays |
Electric current |
Catecholamines and degradation products |
|
7) Separation by charge |
Electrophoresis/HPLC |
Plasma proteins, Hbs |
|
8) Trace elements |
Atomic absorption |
Se, Zn, etc. |
(Source: Schleicher, 2006)
A typical Point-of-Care analysis
process
1.
Insert sample in
microfluidic lab on a chip
2.
Place lab-on-a-chip
cartridge in analyzing instrument
3.
Press start and
wait 5-10 minutes for printout of results
![]()
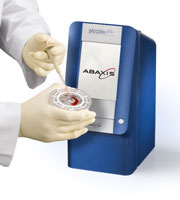


Source: http://www.abaxis.com/medical/piccolo_operation.html
New enabling technology
Technologies
and processes that will further enable POC technology are summarized as follows
1.
Exploiting
scaling phenomena to discover new detection schemes for future applications
2.
Disposables
interfacing with a non disposable housing/analyzer
3.
Taking large
pieces of equipment and shrinking them down
4.
Reducing sample
size and reagents used
5.
Improving
detection sensitivity
6.
Reducing processing
time
7.
Mass parallel
processing
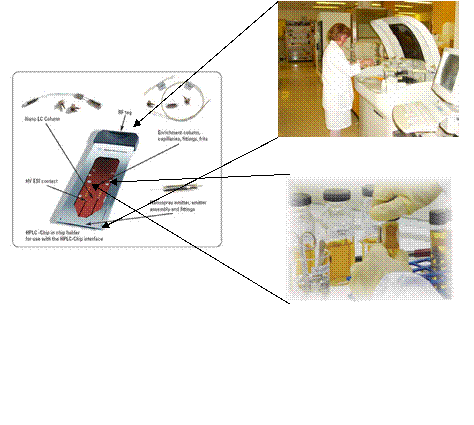
Examples
technologies
What was once a large bulky piece of equipment with large
power consumptions has now been miniaturized into a portable, laptop sized
ultrasound imaging device for emergency response personnel and third world
countries.
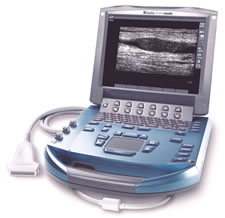
Source:
http://www.idsa.org/IDEA2006/galleries/idea/idea2006/award_details.asp?id=83&cat=9
On this disk we can perform over 24 different tests in
a single process on a disposable disc the size of a CD. All fluid transport is
driven by capillary and centrifugal forces to enable parallel processing of
data on a single run.
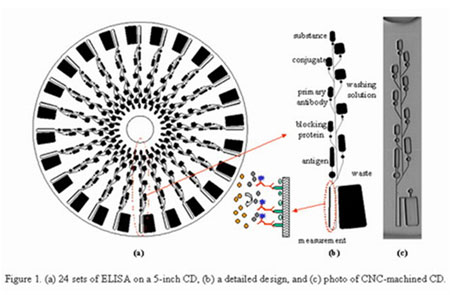
Source: www.bioloc.net/cd-elisa-unique.htm
New technology and detection schemes allow for the
processing and detection of multiple analytes or pathogens within a single test
using an array based immunoassay. Here an electrochemical sensor can detect the
byproducts of different catalysts functionalized by three different types of
antibodies. In this fashion, multiple targets can be detected on a single test
run.
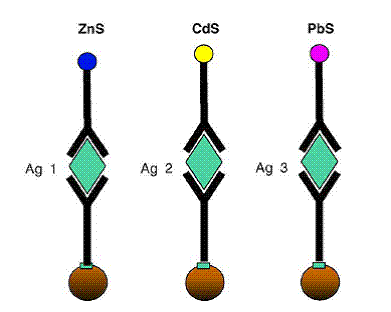
(Source: Wang, 2006)
Here they exploit the scaling effects of a magnetic
field to develop a Hall Effect sensor combined with a sandwich immunoassay
technique.
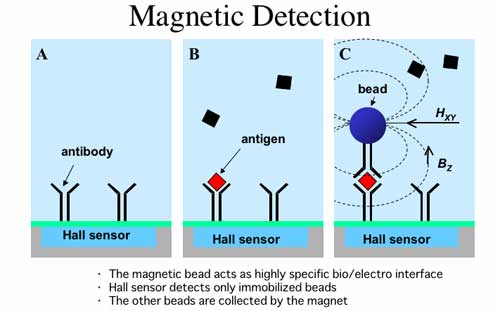
Source: www.coe.berkeley.edu/labnotes/1003/boser.html
A recent POCT Technology Review
For
a thorough analysis and review of the current point of care technology, refer
to the link below.
http://www.prwebdirect.com/releases/2006/5/prweb387007.php
Home General Diagnostics Information
Clinical Laboratory Diagnostics
Current Point-of-Care
Diagnostics Current
Diagnostics Companies Future Point-of-Care
Diagnostics References