



Discovery of Transdermal Penetration Enhancers by High-throughput Screening
Methods
Formulation Screening Methods
* In vitro skin impedance guided high throughput (INSIGHT) Screening
* Developed from a high-throughput method using pig skins for impedance measurements
* Based on the law of diffusion through the stratum corneum (Fick's Law)
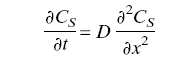
(Cs is the concentration of solutes penetrate the skin. D is diffusion coefficient of solutes in stratum corneum.)
This equation was solved analytically with various boundary conditions to the following form:
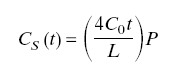
(P is the long-time (steady state) permeability. L is the length of stratum cornea. t is time)
This equation shows that the instantaneous concentration of the solute in the stratum corneum is proportional to the
steady-state permeability of the stratum corneum. Thus, this concept is applied to measure the concentration of solutes
in short-time to screen the efficacy of the combinatorial CPEs.
* Screening apparatus consists of a donor plate (on top made from Teflon) and a receiver plate (poly carbonate) (Fig 6)

Fig6. Receiver and donor plate of the INSIGHT method with 10x10 wells. Between two plates is the pig skin with stratum cornea facing the donor plate. One electrode is attached to the dermal layer, the other is attached to each of the well to measure the conductivity (permeability) of each condition. Each well is filled with different CPEs or SCOPEs.
* Based on measurements of concentrations of radiolabelled (mannitol) solutes into the skin and skin impedance as
a determinant for good SCOPEs candidates.
* Skin impedance, conductivity were measured at 0 and 24 h.
* Conductivity enhancement ratio (ER) was calculated.
* Synergy values determine the combinatorial effect of SCOPEs
S: synergy value; X,Y is percentage of CPE A and B respectively. ER: enhancement ratio
Experimental Design
From the library of CPEs, SCOPEs are generated in pairs, which are screened by High-throughput screening (INSIGHT).
Screened SCOPEs are investigated for irritation potential assessment via EpiDerm. Franz Diffusion Cells (FDC) is
then used to validate the SCOPEs for their efficacy and safety. Lastly, identified SCOPEs are tested in animal models
for bioavailability to determine the successful formulation.
Fig7. The experimental design for INSIGHT method